Question 2#
A 33-year-old alcoholic man is hospitalized for fever, chills, and cough productive of currant jelly sputum. Blood and sputum cultures are drawn, and a chest x-ray is consistent with lobar pneumonia. Urine toxicology screen and a serum ethanol level are negative. After being admitted and started on empiric antibiotics, he continues to decompensate and becomes more hypoxemic based on pulse oximetry.
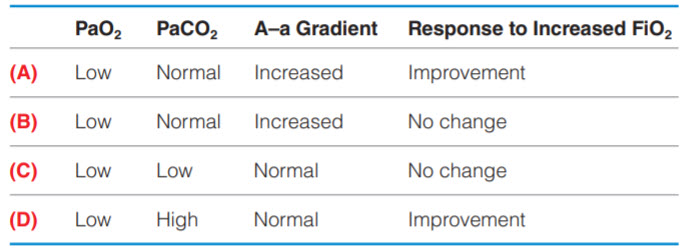
Which of the following choices best represents the pathophysiologic parameters seen in this patient? (Note: A–a gradient is Alveolar– arterial gradient; FiO2 is the fraction of inspired oxygen.)
B. (B)
C. (C)
D. (D)
Correct Answer is B
Comment:
Low PaO2, normal PaCO2, increased A–a gradient, and no change in response to increased FiO2. This question assumes a background knowledge of the physiology of gas exchange and how to differentiate between hypoxemic and hypercapnic respiratory failure. Most cases of hypoxemia involve an elevation in the Alveolar–arterial (A–a) gradient, which is the product of the partial pressure of alveolar oxygen (PAO2) minus the partial pressure of arterial oxygen (PaO2). The A–a gradient normally increases with aging and is represented by the formula: “age/4 + 4.” (Although there is an equation for calculating the PAO2, the concept is more important than the equation for the shelf examination.) There are many causes of hypoxemia, and it is best to use a systematic approach for this discussion.
The causes of respiratory failure can be broken up by whether there is hypoxemia with a low/normal PaCO2 (hypoxemic respiratory failure) or there is hypoxemia and an elevated PaCO2 (hypercapnic respiratory failure). Major causes of hypoxemic respiratory failure include ventilation/perfusion (V/Q) mismatch, right-to-left shunts, and diffusion impairment. V/Q mismatch is a disease process that causes an imbalance between blood flow and gas exchange, which decreases the efficiency of gas exchange and causes hypoxemia. This is really a gray zone in between two states: if taken to one end of the spectrum with a pure loss of perfusion (e.g., pulmonary embolism), this causes “dead space ventilation” (normal air flow but no blood flow). If taken to the other end of the spectrum with a pure loss of ventilation (e.g., atelectasis or pneumonia with consolidation), then this becomes a shunt. Shunts are simply the result of blood bypassing any area of gas exchange, carrying hypoxemic blood back into the circulation. Shunts can also be extrapulmonary (e.g., intracardiac shunts like an atrial septal defect). An example of diffusion impairment is interstitial lung disease with fibrosis that increases the distance that oxygen must traverse to get from the alveoli to the blood stream; processes that increase the rate of blood flow (exercise) or decrease the oxygen-carrying capacity (anemia) can exacerbate hypoxemia caused by diffusion impairment. Other causes besides the above three mechanisms include poisoning (carbon monoxide, methemoglobinemia, and cyanide) and high altitudes (same fraction of inspired oxygen but lower atmospheric pressure). Hypercapnic respiratory failure is caused by inadequate ventilation (respiratory rate × tidal volume), and results from processes such as neuromuscular disease, oversedation, COPD, and obesity hypoventilation syndrome. In hypoxemic respiratory failure, the patient attempts to compensate for the hypoxemia by increasing ventilation, which results in a normal or low PaCO2.
This patient has hypoxemic respiratory failure from pneumonia, and there are no reasons for hypoventilation (no sedation and a negative drug screen). In a process like a lobar pneumonia, where alveoli are completely filled with pus, shunting will predominate over V/Q mismatch and therefore the hypoxemia will not be corrected with increased oxygen administration. Positive end-expiratory pressure (PEEP) might help to open some of the alveoli in areas of consolidation, converting a shunt to a V/Q mismatch, but this question asks about the response to FiO2 alone.